 Copper Contamination in Drinking Water
Copper Contamination in Drinking Water
Corrosive water & copper pipe health hazards
- POST a QUESTION or COMMENT about this article topic.
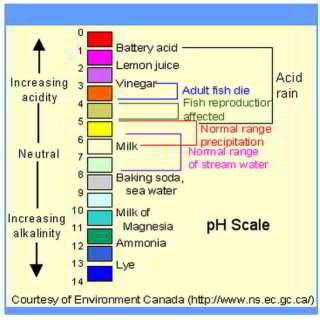
Aggressive, corrosive, low pH water - acidic water can cause high levels of copper in the drinking water supply:
This article discusses the health effects of dissolved copper in drinking water and the relation between aggressive or corrosive water and high levels of copper. We include recommended limits for copper in drinking water.
This article series describes effects of low pH, acidic or corrosive water on building piping, leaks, dissolved copper, health hazards, and the plumbing system in general. We describe how to detect corrosive or aggressive water and what should be done about it.
InspectAPedia tolerates no conflicts of interest. We have no relationship with advertisers, products, or services discussed at this website.
- Daniel Friedman, Publisher/Editor/Author - See WHO ARE WE?
Copper Contamination in Drinking Water, Health Effects & Limits
 Corrosive water is responsible for health effects of increased lead, copper or other contaminants in drinking water and corrosive water in building plumbing & heating systems is also responsible for costly leak damage.
Corrosive water is responsible for health effects of increased lead, copper or other contaminants in drinking water and corrosive water in building plumbing & heating systems is also responsible for costly leak damage.
Health and aesthetic impacts of copper corrosion on drinking water
Traditional research has focused on the visible effects of corrosion--failures, leaks, and financial debits--and often overlooked the more hidden health and aesthetic aspects.
Clearly, corrosion of copper pipe can lead to levels of copper in the drinking water that exceed health guidelines and cause bitter or metallic tasting water.
Because water will continue to be conveyed to consumers worldwide through metal pipes, the water industry has to consider both the effects of water quality on corrosion and the effects of corrosion on water quality.
Integrating four key factors -- chemical/biological causes, economics, health and aesthetics -- is critical for managing the distribution system to produce safe water that consumers will use with confidence.
As technological developments improve copper pipes to minimize scaling and corrosion, it is essential to consider the health and aesthetic effects on an equal plane with chemical/biological causes and economics to produce water that is acceptable for public consumption.
- [15] "Health and aesthetic impacts of copper corrosion on drinking water", Dietrich AM, Glindemann D, Pizarro F, Gidi V, Olivares M, Araya M, Camper A, Duncan S, Dwyer S, Whelton AJ, Younos T, Subramanian S, Burlingame GA, Khiari D, Edwards M., Virginia Tech, Blacksburg, VA 24061-0246, USA. andread@vt.edu, Water Sci Technol. 2004
Recommended Maximum Limits for Copper in Drinking Water
EPA's list of limits on contaminants in water (http://water.epa.gov/drink/contaminants/index.cfm#List) is set in MCL (Maximum Contaminant Level), MCLG (Maximum Contaminant Level Goal) or mg/L (milligrams per liter) (mg/L = ppm) or parts per million - two ways of describing the same concentration
For copper in drinking water the U.S. EPA recommends both a MCLG and an "action level" of 1/3 mg/L for copper in drinking water, citing short term gastrointestinal distress (short term exposure) or liver or kidney damage (long term exposure).
The EPA also warns " People with Wilson's Disease should consult their personal doctor if the amount of copper in their water exceeds the action level"
Test the level of copper in your water - how to construct a "worst case" test
It would be instructive to test your water's copper content to see if it's unusually high. Not only would that tell us if acidic well water is corroding the pipe interior, you might find that the levels of copper are high enough to merit action for water safety / potability.
Making a "worst case" (most critical or "safest") measurement for copper or any other contaminant that might be found in drinking water due to dissolving the contaminant out of the water piping and supply system equipment can be done following the DF approach.
We want to collect a sample of water that is most likely to represent the highest concentration likely to occur in household water that someone might drink.
To construct the "worst plausible case" scenario we collect a water sample after water has been sitting in the pipes for some time period. A longer wait period (days, weeks, months) might represent a house that has sat unoccupied for some time. But in my opinion I'd collect water that has sat overnight as a more likely and thus more plausible "worst case" scenario.
If all of the house plumbing is copper, you would, on "the morning after", go to a plumbing fixture most distant from the incoming water supply, pump, and tank, open a cold (test 1) water faucet and run enough water to flush out the faucet and any plastic fixture risers, so that we're sure we've got water that was in the copper piping overnight.
Then collect the sample.
Watch out: at WATER TESTS for CONTAMINANTS we discuss testing well water quality - what is "in" the well water that may make it unsafe or unpalatable to drink.
Water that may be too high in mineral content, clogging pipes and water heaters
See MEASURE WATER HARDNESS
or water that is too acidic or corrosive, causing leaks in copper piping or that causes green water or stains on plumbing fixtures & laundry
see GREEN STAINS from WATER SUPPLY
...
Continue reading at pH of DRINKING WATER, HEALTH EFFECTS or select a topic from the closely-related articles below, or see the complete ARTICLE INDEX.
Or see these
Recommended Articles
- CORROSIVITY or ACIDITY of WATER
- CORROSIVITY PROBLEMS in WATER SYSTEMS
- CAUSES of CORROSIVE WATER
- COPPER IN WATER, HEALTH EFFECTS
- COPPER LIMITS in DRINKING WATER
- pH of DRINKING WATER, HEALTH EFFECTS
- LANGLIER SATURATION INDEX LSI
- LEAD IN DRINKING WATER, HOW to REDUCE
- LEAK INSPECTION of WATER SYSTEMS
- OTHER CAUSES of PINHOLE LEAKS in COPPER PIPES
- OTHER CAUSES of PIPE PITTING: pH & HCO3 LEVELS
- PINHOLE LEAKS by ELECTRICAL GROUND ERRORS
- WATER TREATMENT EQUIPMENT CHOICES - topic home
- LEAK CAUSES in WATER PIPING
Suggested citation for this web page
COPPER IN WATER, HEALTH EFFECTS at InspectApedia.com - online encyclopedia of building & environmental inspection, testing, diagnosis, repair, & problem prevention advice.
Or see this
INDEX to RELATED ARTICLES: ARTICLE INDEX to WATER TREATMENT SYSTEMS
Or use the SEARCH BOX found below to Ask a Question or Search InspectApedia
Ask a Question or Search InspectApedia
Try the search box just below, or if you prefer, post a question or comment in the Comments box below and we will respond promptly.
Search the InspectApedia website
Note: appearance of your Comment below may be delayed: if your comment contains an image, photograph, web link, or text that looks to the software as if it might be a web link, your posting will appear after it has been approved by a moderator. Apologies for the delay.
Only one image can be added per comment but you can post as many comments, and therefore images, as you like.
You will not receive a notification when a response to your question has been posted.
Please bookmark this page to make it easy for you to check back for our response.
IF above you see "Comment Form is loading comments..." then COMMENT BOX - countable.ca / bawkbox.com IS NOT WORKING.
In any case you are welcome to send an email directly to us at InspectApedia.com at editor@inspectApedia.com
We'll reply to you directly. Please help us help you by noting, in your email, the URL of the InspectApedia page where you wanted to comment.
Citations & References
In addition to any citations in the article above, a full list is available on request.
- De Waard, C., and D. E. Milliams. "Carbonic acid corrosion of steel." Corrosion 31, no. 5 (1975): 177-181.
- Isaac, R. A., L. Gil, A. N. Cooperman, K. Hulme, B. Eddy, M. Ruiz, K. Jacobson, C. Larson, and O. C. Pancorbo. "Corrosion in drinking water distribution systems: a major contributor of copper and lead to wastewaters and effluents." Environmental science & technology 31, no. 11 (1997): 3198-3203.
- Jones, Anne. "Stress corrosion cracking." In in ASM Handbook, Metals Handbook. 1998.
- Kermani, M. B., and A. Morshed. "Carbon dioxide corrosion in oil and gas production—a compendium." Corrosion 59, no. 8 (2003): 659-683.
- Kiene, L., W. Lu, and Y. Levi. "Relative importance of the phenomena responsible for chlorine decay in drinking water distribution systems." Water Science and Technology 38, no. 6 (1998): 219-227.
- Kritzer, Peter. "Corrosion in high-temperature and supercritical water and aqueous solutions: a review." The Journal of Supercritical Fluids 29, no. 1 (2004): 1-29.
- Little, Brenda J., Florian B. Mansfeld, Peggy J. Arps, and James C. Earthman. Microbiologically influenced corrosion. Wiley‐VCH Verlag GmbH & Co. KGaA, 2007.
- Oram, Brian, "Drinking Water Issues Corrosive Water (Lead, Copper, Aluminum, Zinc, and More)" (web page), Water Research Center, Water Research Center B.F. Environmental Consultants Inc. 15 Hillcrest Drive, Dallas, PA 18612, USA, retrieved 2017/02/16, original source: http://www.water-research.net/index.php/drinking-water-issues-corrosive-water-lead-copper-aluminum-zinc-and-more
- Roberge, P. R. (1999). Handbook of Corrosion Engineering (1st ed.). McGraw-Hill Professional. ISBN 0-07-076516-2.
- Sigler, W. Adam, and Bauder, Jim, "CORROSIVITY" [PDF], Montana State University Extension, Water Quality Program, Department of Land Resources and Environmental Sciences, (un-dated) retrieved 2017/02/16, original source: http://waterquality.montana.edu/well-ed/files-images/Corrosivity.pdf - note that this page was adapted from Wilkes University Center For Environmental Quality; Corrosion, Saturation Index, Balanced Water in Drinking Water Systems
- Speller, Corrosion. Causes and Prevention of Corrosion in Steam and Hot Water Heating Systems. McGraw-Hill, 1951.
- Volk, Christian, Esther Dundore, John Schiermann, and Mark LeChevallier. "Practical evaluation of iron corrosion control in a drinking water distribution system." Water research 34, no. 6 (2000): 1967-1974.
- Was, G. S., P. Ampornrat, G. Gupta, S. Teysseyre, E. A. West, T. R. Allen, K. Sridharan et al. "Corrosion and stress corrosion cracking in supercritical water." Journal of Nuclear Materials 371, no. 1 (2007): 176-201.
- [2] Clean Water Systems & Stores, Inc. 2806-A Soquel Ave, Santa Cruz, California 95062, Telephone: 1-888-600-5426 or international: 1-831-462-8500 . web search 4/23/12, original source: - cleanwaterstore.com/copper-pipe-corrosion.html
- [3] "pH in Drinking-water Background document for development of WHO Guidelines for Drinking-water Quality", in Guidelines for drinking-water quality, 2nd ed. Vol. 2. Health criteria and other supporting information, World Health Organization, Geneva, 1996. Web search 4/23/12, original source: http://www.who.int/water_sanitation_health/dwq/chemicals/en/ph.pdf
- [4] "Langelier Saturation Index (LSI), Wikipedia Web: https://www.wikipedia.org/ provided background information about some topics discussed at this website provided this citation is also found in the same article along with a " retrieved on" date. NOTE: because Wikipedia entries are fluid and can be amended in real time, we cite the retrieval date of Wikipedia citations and we do not assert that the information found there is necessarily authoritative.
- [5] "Drinking Water Contaminants, List of Contaminants & their MCLs", U.S. EPA United States Environmental Protection Agency, National Primary Drinking Water Regulations, web search 4/23/12, original source: http://water.epa.gov/drink/contaminants/index.cfm#List
- [6] "Basic Information about Copper in Drinking Water", U.S. EPA United States Environmental Protection Agency, web search 4/23/12, original source: http://water.epa.gov/drink/contaminants/basicinformation/copper.cfm
- [7] "Fin Tube / Bare Elements", Slant/Fin Boilers & Baseboards, Slant/Fin Corporation, 100 Forest Drive, Greenvale, NY 11548, Phone: (516) 484-2600, Fax: (516) 484-5921, E-mail: info@slantfin.com, web search 4/23/12, original source: http://www.slantfin.com/index.php/products/baseboard-residential/fin-tube--bare-elements
- [14] "Pitting Corrosion in Copper Tubes – Cause of Corrosion and Counter-Measures", Mattsson, E.; Fredriksson, A.-M., British Corrosion Journal, Volume 3, Number 5, September 1968 , pp. 246-257(12), Maney Publishing, Quoting the article abstract:
An investigation of failures of hard-drawn copper water pipes (phosphorus-deoxidised copper) in service due to pitting corrosion was conducted from November, 1962 to February, 1965. Fifteen cases were reported. All those about which information could be obtained came from hot water installations and occurred in water with a low pH (?7) and a HCO3- content of, at the most, 100 mg/l but generally below 50 mg/1. Failures not due to pitting corrosion (i.e. caused by erosion and corrosion or corrosion fatigue) occurred in waters with a higher pH and higher HCO3- content.
A laboratory investigation into the ability of the corrosion products to counteract further corrosion in different types of water was also carried out, using an electrolytic cell which, in principle, was a model of an active pit in a copper tube. This led to the following conclusions, which are in good agreement with the results obtained from the examination of service failures:
If the pH value of the water is high enough, the copper dissolved by the corrosion can be precipitated as basic copper salt. At low pH values such precipitation does not take place.
If the [HCO3?]/[SO42?] ratio in the water is high, dissolved copper can be precipitated as basic copper carbonate in the neighbourhood of the corrosion site and counteract further corrosion.
At a low [HCO3?]/[SO42?] ratio, crusts of basic copper sulphate will be precipitated at some distance from the corrosion site and may lead to a high corrosion rate.
Pitting is not likely to occur in hot water tubes of hard copper if the pH is ? 7·4 and the [HCO3?]/[SO42?] ratio ?1 (the concentrations given in mg/1). The critical values mentioned are approximate and may be adjusted in the light of future experience. - [15] "Health and aesthetic impacts of copper corrosion on drinking water",
Dietrich AM, Glindemann D, Pizarro F, Gidi V, Olivares M, Araya M, Camper A, Duncan S, Dwyer S, Whelton AJ, Younos T, Subramanian S, Burlingame GA, Khiari D, Edwards M., Virginia Tech, Blacksburg, VA 24061-0246, USA. andread@vt.edu, Water Sci Technol. 2004;49(2):55-62., Abstract
Traditional research has focused on the visible effects of corrosion--failures, leaks, and financial debits--and often overlooked the more hidden health and aesthetic aspects. Clearly, corrosion of copper pipe can lead to levels of copper in the drinking water that exceed health guidelines and cause bitter or metallic tasting water. Because water will continue to be conveyed to consumers worldwide through metal pipes, the water industry has to consider both the effects of water quality on corrosion and the effects of corrosion on water quality. Integrating four key factors--chemical/biological causes, economics, health and aesthetics--is critical for managing the distribution system to produce safe water that consumers will use with confidence. As technological developments improve copper pipes to minimize scaling and corrosion, it is essential to consider the health and aesthetic effects on an equal plane with chemical/biological causes and economics to produce water that is acceptable for public consumption. - Clogged Drain Diagnosis: Is it a blocked drain or the septic system? - A First Step for Homeowners
- CROSS CONNECTIONS, PLUMBING - bacterial contamination of building piping - sources
- Plastic Water Supply & Drain Piping Failures, Leaks, Repairs, Lawsuits: Water Supply Piping Defects and Plastic or polybutylene water supply piping failures, leaks, and litigation.
- PLASTIC PIPING ABS CPVC PB PEX PPR PVC - Leaks, Repairs, Lawsuits: Water Supply Piping Defects and Plastic or polybutylene water supply piping failures, leaks, and litigation. Our main article on plastic supply and drain piping leaks, failures, litigation, repairs, at PLASTIC PIPING ABS CPVC PB PEX PPR PVC. Excerpts are just below.
- PB (polybutylene with mechanical fittings) Piping leaks: especially in mobile homes and in the U.S. South, for example trailers and double-wides throughout Florida that were piped with this material - per M Cramer. See "Polybutylene Piping: Time Bomb?" Daniel Friedman, Journal of Light Construction, August 1996 [Technical Q&A] .
- In addition to citations & references found in this article, see the research citations given at the end of the related articles found at our suggested
CONTINUE READING or RECOMMENDED ARTICLES.
- Carson, Dunlop & Associates Ltd., 120 Carlton Street Suite 407, Toronto ON M5A 4K2. Tel: (416) 964-9415 1-800-268-7070 Email: info@carsondunlop.com. Alan Carson is a past president of ASHI, the American Society of Home Inspectors.
Thanks to Alan Carson and Bob Dunlop, for permission for InspectAPedia to use text excerpts from The HOME REFERENCE BOOK - the Encyclopedia of Homes and to use illustrations from The ILLUSTRATED HOME .
Carson Dunlop Associates provides extensive home inspection education and report writing material. In gratitude we provide links to tsome Carson Dunlop Associates products and services.

