 Mothball Odor Hazards
Mothball Odor Hazards
Health risks of Mothballs: naphthalene / p-dichlorobenzene; removal of naphthalene odors
- POST a QUESTION or COMMENT about mothball chemicals, hazards, odors, & odor removal
Mothball or naphthalene odor removal & mothball chemical & gas hazards:
The US EPA estimates that about 7.5 million pounds of naphthalene are marketed in the U.S. each year as a pesticide, of which the major use is in moth repellant products. (US EPA 2008).
Here we describe the detection of and risks of exposure to mothball chemicals & odors when moth repellent products are applied indoors in buildings.
In this article series, we describe how to get rid of mothball odors in buildings, building furnishings, clothing, or vehicles.
In this article series, we also provide research explaining the health hazards from exposure to mothball odors (and gases) as well as the child hazard of eating mothballs or moth repellent products. We discuss methods to reduce mothball chemical & gas exposure.
We cite authoritative sources of information about safe and proper use of moth repellents and about mothball and moth repellent chemistry, child hazards, indoor air quality and health concerns, and proper application and use of these products.
Page top photo, courtesy of Wikipedia commons, shows a cluster mothballs, or a solid form of naphthalene.
InspectAPedia tolerates no conflicts of interest. We have no relationship with advertisers, products, or services discussed at this website.
- Daniel Friedman, Publisher/Editor/Author - See WHO ARE WE?
Mothball Smell & Chemicals, Proper Use, Hazards, Exposure, Health Effects, Odor Removal
 Mothballs, like those shown at the top of this page, are fumigants containing naphthalene
or paradichlorobenzene and act as a pest repellent and possibly act also as a pesticide used to protect clothing and other soft goods from attack by clothes moths (Tineola bisselliella or the casemaking clothes moth Tinea pellionela)
Mothballs, like those shown at the top of this page, are fumigants containing naphthalene
or paradichlorobenzene and act as a pest repellent and possibly act also as a pesticide used to protect clothing and other soft goods from attack by clothes moths (Tineola bisselliella or the casemaking clothes moth Tinea pellionela)
If you are looking for information on how to get rid of mothball odors, go directly to
GET RID of MOTHBALL ODORS - separate article
Mothballs, moth flakes, crystals, and bars are insecticides that are formulated as solids. As such, mothballs are registered as pesticides because they contain high concentrations of one of two active ingredients—naphthalene or paradichlorobenzene (sometimes referred to as 1,4-dichlorobenzene).
Through sublimation, they exude gas, acting as a fumigant. - (Fishel 2017)
[Click to enlarge any image]
Naphthalene has been used as insecticide world wide since about 1880 (Pyenson 1936), but its health effects, also discussed here, were studied a bit later, beginning around 1900 (Nash 1903).
"Clothes moths" Tinea & Tineola sp. (Family: Tineidae), like that shown in this illustration from "Clothes Moths & Carpet Beetles" (USDA 1953), are small 1/2-inch beige or buff colored moths.
Clothes moths are yellowish-tan to buff-colored with a wingspread of about 1/2 inch. The larvae are white with brown to black heads, and are also about 1/2 inch long. (Kass 1981).
Clothes moths attack animal fibers such as wool, felt, and silk, but they may also attack feathers, furs, and even leather. Those materials contain a fibrous protein (keratin) that provides food for clothes moth larvae.
Article Contents
- HOW TO USE MOTHBALLS or MOTH FLAKES PROPERLY
- HEALTH EFFECTS of EXPOSURE to MOTHBALLS
- REDUCE INDOOR EXPOSURE to MOTHBALL CHEMICALS
- LIFE EXPECTANCY of MOTHBALLS & MOTHBALL ODOR
- DIFFERENCE BETWEEN PARADICHLOROBENZENE & NAPHTHALENE MOTHBALLS
- MOTHBALL USE GUIDES from MANUFACTURERS & EXPERTS
How to Use Mothballs, Moth Bars, or Moth Flakes Properly
Naphthalene or paradichlorobenzene fumigants are said by manufacturers of mothballs or moth-flakes to kill or at least deter moths and caterpillars.
Really? Some sources, such as the U.S. California state department of Pesticide Regulation, say that mothballs, in fact, do not repel rats, mice, nor moles.
In addition, some people try using these products to repel other animals or pests even though that use is not recommended and may not be permitted.
However, the use of naphthalene as an animal pest repellant was included in the US EPA's description of that material in its 2008 Registration Eligibility document cited below. (US EPA 2008)
Also see REPELLENTS for ANIMAL PESTS
Below, we provide links to reference documents on how to use mothballs safely. Here, excerpting from those sources we list some basic steps in using mothballs.
 Photo: a common clothes moth Tineola bisselliella - photo source & these moths are cited & described below.
Photo: a common clothes moth Tineola bisselliella - photo source & these moths are cited & described below.
- Identify the pest correctly before using any pesticide.
Mothballs, moth bars, moth flakes are intended only for use in repelling or killing clothes moths like the adult moth shown here. (More photos of clothes moths, clothes moth larvae, and other moths that are not clothes moths are shown on this page.)
Clothes moths are small, 1/2-inch moths that are beige or buff-colored. They have narrow wings that are fringed with small hairs. (Potter )
Photo: common clothes moth, adult form. (Leillinger, cited below) [Click to enlarge any image]
- Don't use mothballs unnecessarily.
Mothballs and their sister products are not needed to protect clothing that is worn or used frequently.
They are used to protect clothing that is to be stored for a protracted interval.
There are alternative means to control clothes moths or to protect clothing as we cite below in this section. (Stone 2008) (Potter) - Read & follow the mothball manufacturer's directions.
Watch out: if you are using mothballs or moth flakes to control or prevent damage from clothes moths, it is essential that you read and follow the product manufacturer's instructions.
Most of the complaints we've received from readers over the past two decades are traced to having used mothballs improperly. - Use mothballs only in an air-tight container such as a garment bag or well-sealed plastic container.
Typical mothball label instructions instruct the user to place the mothballs, moth flake, or moth bar in a tightly-closed container that will prevent the mothball pesticide fumes from entering building spaces where the fumes might be inhaled by people, pets, or other animals.
When contained in the air-tight container the mothball fumes accumulate at a concentrated level and thus can kill clothes moths or clothes moth larvae.
Do not put mothballs into an open container, nor into a simple plastic garbage bag.
- Never mix moth-repellent products that use different ingredients: don't mix naphthalene (often misspelled as napthalene) with paradichlorobenzene.
- Never distribute mothballs or moth flakes in open space nor into building cavities.
Do not distribute mothballs in an open space such as a closet or basement or attic floor nor inside a care or other vehicle.
Do not place mothballs in wall cavities nor crawl spaces nor any other building cavity.
Do not place mothballs or moth flakes outdoors such as in garden areas in an effort to repel other animals. There the mothballs can harm children or pets, their us in that location is illegal; paradichlorobenzene or naphthalene can contaminate soil, plants, or water.
Otherwise you may find that the mothball odor can persist and can be very difficult to get rid-of.
- Never use mothballs or moth-flakes where young children or pets may come near them.
Naphthalene can damage a child's red blood cells and is toxic to all animals including children or pets. - Do not use moth balls or flakes in any location or application that is prohibited by the manufacturer's label or instructions.
- Below on this page see mothball use detailed instructions in these
MOTHBALL USE GUIDES from MANUFACTURERS & EXPERTS
In the U.S., the EPA permits use of moth balls to repel moths and caterpillars only. Because moth balls are toxic to humans and pets, mothballs are not permitted for use in buildings to repel animal pests like squirrels or bats.
Watch out:Some people have also tried using moth repellent products like mothballs to keep rodents and other pests out of certain building areas or out of stored vehicles.
Placing an open box of mothballs under the hood of my MG Midget while it was stored kept squirrels and mice from nesting in the engine compartment, and leaving a box of mothballs inside the car was an attempt to keep the same pests out of the vehicle interior, too.
Watch out: we do not recommend placing mothballs, moth cakes, or moth repellent flakes inside building rooms, crawl spaces, attics, wall or ceiling cavities, trash cans, nor inside vehicles, both because the fumes and chemicals can be hazardous to humans and because the odor can later be difficult to get rid of. We explain this problem in detail below.
Watch out: because mothballs are a registered pesticide and because of the toxicity of their chemicals, it is illegal to use mothballs or moth crystals, cakes, etc. as a repellent for animal pests (birds, cats, deer, dogs, moles, pigeons, mice, skunks, raccoons,snakes, squirrels, etc.) [19]
The clothes moth image shown just below is from Stone & Stock, PNW who provide an excellent guide to moth control. [19]
 As stated
at PESTICIDE EXPOSURE HAZARDS
As stated
at PESTICIDE EXPOSURE HAZARDS
Pesticides are a special class of organic chemicals designed to kill living organisms.
In addition to the compounds used in the home and garden, the class of chemicals regulated as pesticides also include kitchen and bath disinfectants, flea and tick products, and swimming pool chemicals.
In most cases, both the active ingredient targeted to one or more pests and the “inert” carriers are organic chemicals that are toxic to humans.
Every registered pesticide has a “signal” word on the label, ranking the level of toxicity to humans, as follows:
- Danger—Poison: highly poisonous
- Danger: poisonous or corrosive
- Warning: moderately hazardous
- Caution: least hazardous
Health Effects of Exposure to (or Ingestion of) Mothballs, Moth cakes, Moth Repellent Crystals
 Mothballs contain one of the chemicals, paradichlorobenzene or naphthalene. Paradichlorobenzene is classified as a possible human carcinogen by the EPA, and its vapors can irritate skin, eyes, and the respiratory tract.
Mothballs contain one of the chemicals, paradichlorobenzene or naphthalene. Paradichlorobenzene is classified as a possible human carcinogen by the EPA, and its vapors can irritate skin, eyes, and the respiratory tract.
Large doses can damage the liver. Mothballs are not intended to be placed in open spaces such as rooms, closets, or vehicles. Rather they should be used in an airtight space such as a clothes storage bag. [7]
The moths shown here are not clothes moths. The insects shown are attacking our fig plant but not our clothing. - Ed.
Mothballs are fumigants that will dissolve or sublime at lower temperatures; mothballs thus work by a process of sublimation - the solid ball of chemicals converts directly to a gas that enters the air nearby. [2][7]
But believe it or not, mothballs or moth crystals may also be a child hazard if eaten - as has happened. [3][6][7]
Symptoms of exposure to naphthalene include headache, nausea, dizziness, and difficulty breathing. Paradichlorobenzene is also a potential hazard, although typically less so compared to naphthalene...
Eating just one mothball containing naphthalene can damage a young child’s red blood cells.... [7]
Exposure to naphthalene promotes hemolytic anemia, associated with fatigue in mild cases and acute kidney failure in severe cases. Poisonings of infants have been reported after dressing the children in clothing stored in naphthalene mothballs.
Below: an edited (excerpted) page from a 1963 Boys Life magazine describing a do-it-yourself home science project using moth balls, water, baking soda, and vinegar.
Watch out: we do not recommend repeating this experiment unless under supervision of your science teacher or another suitable expert.
Mothballs are a particular hazard to children, especially small children who may mistake a mothball for something edible.
FYI for our Australian readers, there have been recent news reports inquiring if mothballs are harmful to babies - we're researching that question.
Certainly eating or sucking on mothballs, and probably exposure to high levels of mothball offgassing can be harmful to babies, and mothball odors can trigger problems for people with allergies and asthma (including this site editor). (Sudakin 2011).
Dawson, back in 1958, documented this concern: it's certainly not "new" news.
Watch out: at TERMITES & NAPHTHALENE HAZARDS we point out that there can also be naturally occurring sources of naphthalene in buildings. These, too, might be at a sufficient concentration to pose a health hazard.
Watch out: See details about mothball or naphthalene odor hazards
at NAPHTHALENE POISONING SYMPTOMS
How to Reduce Indoor Exposure to Mothball Odors, Chemicals, Gases
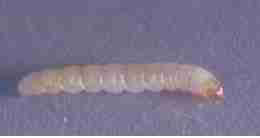
- Avoid unnecessary use of mothballs & moth repellents.
When possible, the best approach is to find non-chemical approaches to pests, including moths. That includes- Don't use moth repellents if you don't have a moth problem in the first place.
Moth larvae are shiny light colored wormlike organisms about 1/2-inch in length. Adult clothing-eating moths are white.
(Image at left, PNW) [19]
To check for a moth infestation in your home, and keeping in mind that most moths are not the species that attack fabrics, you can try a pheromone trap to monitor for moth activity.[2] - Avoid importing moths, moth pupae or moth larvae (it's the larvae that eat animal fibers and fabrics) by cleaning used or old clothing that is "new" to your home before storing those materials away
- When storing clothing or other soft goods, especially woolens or other animal fiber materials, use clothing storage containers that are dry and airtight, first to keep moths out, and second, especially if moth repellent products are to be placed therein, to prevent mothball odors from invading other building areas or materials.
- Do not simply place moth repellent products in open boxes or loose in areas such as rooms, closets, garages, or vehicles nor into building cavities such as ceilings, attics, walls, crawlspaces in an attempt to keep them moth (or mouse, or squirrel) free.
- Cedar closets? Cedar fumes are toxic to moths only at high concentrations. Don't trust a cedar closet alone to prevent moth damage, although indeed, cedar wood and cedar oils do avoid the mothball hazard and odor issue.[2]
- Dried lavender and cedar fiber balls as moth repellents are not supported by good evidence. [2-Farr]
- Don't use moth repellents if you don't have a moth problem in the first place.
- Wash or dry clean clothing before putting it into storage; clean clothes are less attractive to moths according to some experts. Moth eggs and moth larvae, as well as adult moths, can all be killed by a hot water wash cycle.
Moth eggs, pupae, or even adults may already be present in clothing, including cotton, even though moths are not directly attracted to that material. Dry-clean wool or other animal fiber materials (to which moths are attracted) for the same reason. [2]
Shown here: older "moth-proofing", moth blocking, and moth repellent products from Fuller Products and Avon Products.
- Read the mothball package label and closely follow instructions. If you are going to use a moth repellent product like mothballs, read and follow the directions on the product label.
Watch out: keep these products away from children who may be especially vulnerable not only to vapors from mothballs but as something sometimes eaten.
Watch out: also, some of the moth proofing products shown here are self-described as flammable and must be used only as directed.
- Dispose of unwanted mothballs safely. Most of these chemicals contain VOCs that will vaporize and get into the household air. If you cannot dispose of partially used containers, store outside the living space.
- Minimize exposure to moth repellant products. When used, place mothballs, moth repellent cakes or moth crystals in a well-sealed trunk or other container that can be stored in ventilated areas outside of the main living space, such as attics or attached garages.
- Minimize exposure to some air fresheners. Paradichlorobenzene is also the active ingredient in many air fresheners and should be avoided.
Life Expectancy of Mothballs Used in Buildings, Mothball Sublimation Rate, Mothball Odor Persistence
 On 2017-07-27 by Laurie Davis - What is the life expectancy of a moth ball in a hot area?
On 2017-07-27 by Laurie Davis - What is the life expectancy of a moth ball in a hot area?
What is the life expectancy of a moth ball in a hot area? How long does it continue to release the harmful fumes?
On 2017-07-27 by (mod) - Life Expectancy of Mothballs Used in Buildings - Mothball Sublimation Rate
Laurie,
If you will permit me to wave my arms about mothball sublimation rates as a preface to giving an opinion about how long a mothball will "last" in use, then my estimates of mothball life will, perhaps, make more sense.
Tennakone (1978) looked at the sublimation rate for mothballs - but I've not yet gotten a copy of the full article to support the following opinion with science.
Mothballs exposed to the open air will be seen to shrink in size, eventually disappearing, following a process called sublimation: the solid (mothball) converts to a gas without ever entering an intermediate liquid state.
The rate of sublimation of mothballs is determined principally by the amount of air available to or circulating around the mothball.
That's because the sublimation rate of mothballs depends on the relative naphthalene gas or para-dichlorobenzene vapor pressure difference between the thin layer of air at the surface of the mothball and the surrounding air.
When mothballs are kept in a small sealed container or relatively-airtight bag (as they are in a sealed box of mothballs in the store), the gas vapors are trapped around the mothballs, slowing their rate of sublimation to almost nothing.
But when the same mothballs are spread about in more open air, the vapor pressure differences of the mothball gases permit them to continue to off-gas into the surrounding area.
Mothballs that are actually in the spherical form, about 1/2" in diameter when new, will last from a month to several years or longer depending on the type of mothball and how they are placed.
 Furthermore, the life of mothballs depends on the product chemistry: naphthalene mothballs take much longer to dissipate than mothballs comprised of para-dichlorobenzene.
Furthermore, the life of mothballs depends on the product chemistry: naphthalene mothballs take much longer to dissipate than mothballs comprised of para-dichlorobenzene.
Photo: A moth photographed by the author on the Osa Peninsula, Costa Rica, in 2018. This is NOT a clothes moth.
For example, based on simple experiments and observations of mothballs I've placed in buildings, closets, bureaus, or kept in closed packages, I estimate that:
- a single para-dichlorobenzene mothball in open air may dissipate in a month or two
- a single para-dichlorobenzene mothball placed under clothing and not in open air may dissipate in 3-6 months
- a box of para-dichlorobenzene mothballs left together in the box with the lid cut open and placed in a closet or under clothing may last for more than a year.
- a single naphthalene mothball in open air may dissipate in a 3-6 months
- a single naphthalene mothball placed under clothing and not in open air may dissipate in 12 months
- a box of naphthalene mothballs left together in the box with the lid cut open and placed in a closet or under clothing may last for several years or longer.
- sealed packages of mothballs of either type probably have a shelf life of several years
In a hot area like an attic, where convection currents may be moving more air about, those mothball life expectancies will be less due to the effects of both temperature and air movement or air velocity on the sublimation rate of the mothballs of either formula. Moisture might also be a factor in the the rate of mothball sublimation.
"In air, the half-life of naphthalene and paradichlorobenzene is less than one day and about 31 days, respectively." (Fishel 2014) [1] atReferences or Citations .
But that half-life expression is misleading. It would only be accurate if we could obtain a measured volume of these two gases ALONE, that is, without a source that continues to emit them, and if we could then obtain more specific information about the concentration of those gases in the space and the rate of fresh air ventilation of the space as well as quite a few other factors.
Watch out: however, the fumes from these gases may permeate and remain in clothing or building materials after the actual mothballs or moth crystals have dissipated.
Differences Between Paradichlorobenzene Mothballs & Naphthalene Mothballs
Above are two different moth ball products sold by Enoz®. Enoz® Paradichlorobenzene moth balls and Enoz® "old fashioned" or naphthalene mothballs.
These two mothball substances are chemically different, having different properties.
Naphthalene mothballs produce an off-gassing (through sublimation) that creates an environment that repels moths as well as some other creatures. Naphthalene mothballs, and the odor they produce, remains in the environment of use longer than Paradichlorobenzene mothballs.
That is, it will take longer for the Naphthalene odor to dissipate even when the mothball source has disappeared consumed or been removed.
Paradichlorobenzene mothballs produce less noticeable odor (to humans) and its gas will dissipate in free air more quickly than the offgassing product of Naphthalene mothballs.
According to Enoz®, "Para" mothballs for clothing are approved by the Environmental Protection Agency and sport EPA registration number: 1475-39
If all other parameters are the same (air movement, temperature, etc), Paradichlorobenzene mothballs produce a higher concentration of gas in the treatment area than Naphthalene mothballs, possibly increasing the effectiveness of Paradichlorobenzene for the death or at least the discouragement of moths.
Mothball Use & Instructions & Moth Damage or Infestation Prevention
- CA, MANAGING CLOTHES MOTH INFESTATIONS [PDF] Los Angeles County Health Department, retrieved 2022/-9/01 original source: http://publichealth.lacounty.gov/eh/docs/safety/managing-clothes-moth-infestations.pdf
Excerpt:
The webbing clothes moth (Tineola bisselliella) is worldwide in distribution and is often referred to as the most common clothes moth in the United States. When at rest, the adult moth has a length of 7-8 mm, the wings are a pale golden color without markings, and the head bears a fluffy fringe of copper-colored hairs.
The casemaking clothes moth (Tinea pellionella) is about 6 mm in length when the wings are folded, and has a brownish hue. There are three dark spots on the wings, but the markings may become indistinct as the moth ages.
This moth is not as economically important as the webbing clothes moth in most areas of the United States, but an increased incidence of infestations in Los Angeles County suggests it may soon become a serious pest. This enhanced presence may be a consequence of the heightened importation of woolen goods from abroad. - Dellinger, Theresa A., & Eric Day, CLOTHES MOTHS [PDF] (2021) Virginia Cooperative Extension, retrieved 2022/08/29, original source: https://www.pubs.ext.vt.edu/content/dam/pubs_ext_vt_edu/3104/3104-1576/ENTO-471.pdf
- Enoz BioCare Clothes Moth Trap [illustrated above] - we have not located a safety data sheet SDS for this product.
Website excerpt:
Safe for use in food-handling and food storage areas.
Enoz's manufacturer, Willert, USA 4044 Park Avenue St. Louis, Missouri 63110 United States of America Tel: (+1) 800-325-9680 Email: whp@willert.com Web: https://enozhome.com/product/old-fashioned-moth-flakes/
Willert-China: Willert Home Products Sales (Shanghai) Co., Ltd. Room 2603-2606, Lane 166 Minhong Road Zhonggeng Global Creative Center Minhang District Shanghai 201100 China T: (086) 21 5416 2786
Willert produces Enoz Moth Cake and Enoz BioCare Clothes Moth Traps (pesticide free, pheromone attracts male moths, the company suggests pairing with Enoz Moth Balls), and Enoz No-Zone animal repellent stations [ingredients identification needed - Ed.]
- ENOZ OLD FASHIONED MOTH BALLS SAFETY DATA SHEET [PDF] (2015) - source: home depot.
- ENOZ PRODUCT LIST & MATERIAL SAFETY DATA SHEET [PDF] (2011)
Excerpt: All products contain p-dichlorobenzene as the active ingredient. [ p-dichlorobenzene (1,4-dichlorobenzene) ]
Excerpt: White balls, blocks, crystals, nuggets or tablets. Some products may be encased in paper bags (packets) or plastic containers and some products contain fragrance. Harmful if swallowed or inhaled. Dust and vapors may cause eye and respiratory tract irritation. Flammable.
- ENOZ NO ZONE (stations and refills) Animal Repellent Station SAFETY DATA SHEET [PDF] (illustrated above) Primary ingredient: Naphthalene
Excerpts: This chemical is considered hazardous by the 2012 OSHA Hazard Communication Standard (29 CFR 1910.1200).
Personal Precautions Avoid contact with eyes. Ensure adequate ventilation. Use personal protective equipment.
Evacuate personnel to safe areas. Refer to Section 8 Avoid contact with skin, eyes and clothing. Keep people away from and upwind of spill/leak.
Do not breathe vapors, mist or gas
Product Information Product does not present an acute toxicity hazard based on known or supplied information.
Inhalation There is no data available for this product. Aspiration into lungs can produce severe lung damage. May cause pulmonary edema Pulmonary edema can be fatal .
Watch out: OPINION: because its active ingredient is naphthalene (moth balls),using this product, even as directed by the manufacturer, may create problematic indoor mothball odors and may be unsafe for human or pet or other animal exposure.
- [1] Fishel, Fred, "The FACTS ABOUT MOTHBALLS", [PDF], PI254, Agronomy Department, UF/IFAS Extension, (2014), Fred Fishel, professor, Agronomy Department, and director, UF/IFAS Pesticide Information Office; UF/IFAS Extension, Gainesville, FL 32611, Website: http://edis.ifas.ufl.edu retrieved 2017/07/17, original source: http://edis.ifas.ufl.edu/pi254
- Fishel, F. M. 2005. Evaluation of Pesticides for Carcinogenic Potential. Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://edis.ifas.ufl.edu/pi074
- Fishel, F. M. 2006. Homeowner’s Guide to Pesticide Safety. Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://edis.ifas.ufl.edu/pi051
- Hodgson, Erin W., Jessie L. Trina, Alan H. Roe, CLOTHES MOTHS [PDF] Utah State University, retrieved 2022/09/0,1 original source: https://digitalcommons.usu.edu/extension_curall/877/
Excerpts:
Adult clothes moths or “millers” are harmless and do not cause any damage to fabrics. Larvae can feed on wool, feathers, fur, hair, cotton and synthetic fibers.
Clothes moths prefer darkness and are weak flyers.
Good housecleaning is vital to prevent damage from clothes moths; vacuuming can help prevent clothes moth damage. - Kass, Carolyn, CLOTHES MOTHS [PDF] (1981) Cornell University Insect Diagnostic Library, retrieved from Maine Government data, 2022/08/29 original source: https://www.maine.gov/dacf/php/gotpests/bugs/documents/clothes-moths-cornell.pdf - illustration above is excerpted from this document.
Excerpt:
Watch out: This publication contains pesticide recommendations. Changes in pesticide regulations occur constantly and human errors are still possible. Some materials mentioned may no longer be available and some uses may no longer be legal.
All pesticides distributed, sold or applied in New York State must be registered with the New York State Department of Environmental Conservation (DEC). Questions concerning the legality and/or registration status for pesticide use in New York State should be directed to the appropriate Cornell Cooperative Extension Specialist or your regional DEC office.
READ THE LABEL BEFORE APPLYING ANY PESTICIDE.
Above photos of adult clothes moths cited in the article just below: The adult forms of: left - the common clothes moth (Tineola bisselliella photo by Olaf Leillinger, CC BY-SA 2.5, via Wikimedia Commons); and below, the case-bearing clothes moth or casemaking clothes moth (Tinea pellionela) (photo by Malcolm Storey, CC BY-NC-SA 4.0, via BioImages)
- London Natural History Museum, CLOTHES MOTHS IDENTIFICATION GUIDE [PDF] The Natural History Museum, London, U.K., - retrieved 2022/08/29 original source: https://www.nhm.ac.uk/take-part/identify-nature/common-insect-pest-species-in-homes/clothes-moths-identification-guide.html
Excerpt:
The two moths most commonly found in UK homes doing damage to natural fibres are the common clothes moth (Tineola bisselliella) and the case-bearing clothes moth (Tinea pellionella).
It is the immature larvae, rather than the adult moths, that cause the damage to natural fibres such as woollen clothing, upholstery, carpets and furs. Both moths have a similar life cycle and infestations can be controlled using the same approach.
- Oakes, JF, PRO-PEST CLOTHES MOTH TRAP INSTRUCTIONS [PDF] Manufacturer: Russell IPM Ltd
45 First Avenue, Deeside
Industrial Park, Deeside,
Flintshire, CH5 2NU, United
Kingdom
Tel #: (+44) 1244 281 333
Fax #: (+44) 1244 281 878
Email: info@russellipm.com
Web: www.russellipm.com - US Supplier: J. F. Oakes Sales & Marketing, LLC
Physical: 646 E Fifteenth Street
Mailing: P.O. Box 115
Yazoo City, MS 39194
U.S.A.
Tel #: 662 746 7276
Fax #: 662 746 4568
Email: sales@jfoakes.com
Web: www.jfoakes.com
- original source: http://jfoakes.com/GENINFO/51PCM1001
This trap uses a pheromone that specifically attracts Webbing Clothes Moths - Tineola bisselliella. - Oakes, JF, PRO-PEST CLOTHES MOTH TRAP SAFETY DATA SHEET [PDF] Op. Cit.,
- original source: http://www.jfoakes.com/SDS/051-PRO-CM-2-XX.pdf
Website excerpt: The Pro-Pest R.T.U. Clothes Moth Traps are pre-baited with Tineola bisselliella pheromone to attract the Webbing Clothes Moth. Tineola bisselliella is a major pest in clothing and textiles.
MSDS Excerpt: Active Ingredient: Nature identical pheromones of (E,Z)-2,13-Octadecadienal and (E)-2- Octadecenal
- Potter, Michael F., ENTFACT-609 CLOTHES MOTHS [PDF] University of Kentucky College of Agriculture, department of Entomology, retrieved 2022/08/29, original source: https://entomology.ca.uky.edu/ef609
Excerpt:
Clothes moths are pests that can destroy fabric and other materials. They feed exclusively on animal fibers, especially wool, fur, silk, feathers, felt, and leather.
These materials contain keratin, a fibrous protein that the worm-like larvae of the clothes moth can digest. (In nature, the larvae feed on the nesting materials or carcasses of birds and mammals.)
Cotton and synthetic fabrics such as polyester and rayon are rarely attacked unless blended with wool, or heavily soiled with food stains or body oils.
Serious infestations of clothes moths can develop undetected in dwellings, causing irreparable harm to vulnerable materials. - [7] Stone, David L. (David Louis), Stock, T. (Tim), "MOTHBALLS: PROPER USE AND ALTERNATIVE CONTROLS FOR CLOTHES MOTHS", [PDF] PNW 606-E, May 2008, Oregon State University. Extension Service
Washington State University. Extension
University of Idaho. Extension, May, 2008, retrieved 9/22/12, original source: http://scholarsarchive.library.oregonstate.edu/
xmlui/bitstream/handle/1957/20800/pnw606-e.pdf?sequence=1, citation: http://hdl.handle.net/1957/20800,
Abstract: In some homes, clothes moths can damage garments and other belongings. There are two common species of clothes moths in the Pacific Northwest: the webbing clothes moth (Tineola bisselliella) and the casemaking clothes moth (Tinea pellionella).
The larvae, or immature form, of the moths are responsible for the damage done to personal belongings.
- SpringStar CLOTHES MOTH TRAP MATERIAL SAFETY DATA SHEET [PDF] - SpringStar, (425) 487-4360
P.O. Box 2622
Woodinville, WA
USA 98072
USA Email: info@springstar.net
Web: www.springstar.net This MSDS pertains to: INSECT TRAPS, PHEROMONE-BAITED: (Flour Moth Trap, Flour/Pantry Moth Trap, Pantry Moth Lure,
Bird Seed Moth Trap, New Flour/Pantry Moth Trap, Codling Moth Trap, Clothes Moth Trap)
Excerpt: None of the ingredients of this material meet the definition of "Hazardous Chemical" given in OSHA Hazard Communication Regulation 29 CFR 1910.1200(c).
- USDA CLOTHES MOTHS and CARPET BEETLES - HOW TO COMBAT THEM [PDF] (1966), U.S. Department of Agriculture, retrieved 2022/08/29, original source: https://naldc.nal.usda.gov/download/CAT87213856/pdf - this copy supercedes the 1961 and 1957 editions included below.
- USDA CLOTHES MOTHS and CARPET BEETLES - HOW TO COMBAT THEM [PDF] (1961) U.S. Department of Agriculture, retrieved 2022/08/29, original source: https://naldc.nal.usda.gov/download/CAT87214866/pdf
- USDA CLOTHES MOTHS and CARPET BEETLES - HOW TO COMBAT THEM [PDF] (Original) (1953, 1957), U.S. Department of Agriculture, retrieved 2022/08/29, original source: https://naldc.nal.usda.gov/download/CAT40000325/pdf
- USDA INSECT DAMAGE to CLOTHING, FURS, CARPETS, FURNITURE [PDF] (1965) USDA, The Yearbook of Agriculture 1965 pp. 379-382
- Wright, Susan, PREVENTING DAMAGE from CLOTHES MOTHS & CARPET BEETLES [PDF] College of Agriculture, Consumer and Environmental Sciences New Mexico State University, Guide C-504, - retrieved 2022/09/01, original source: https://pubs.nmsu.edu/_c/C504/
Get Rid of or Remove Mothball Odors
Moved to GET RID of MOTHBALL ODORS
Mothball & Other Odors' Impact on Real Estate Sales - Naphthalene / Paradichlorobenzene (PDCB) Odor Research
This discussion has moved
to MOTHBALL ODOR IMPACT on REAL ESTATE SALES
Symptoms of Naphthalene Poisoning
These mothball or naphthalene poisoning symptoms are now found
at NAPHTHALENE POISONING SYMPTOMS - separate article
Health Hazards of Breathing Mothball Offgassing or of Eating Mothballs: Naphthalene Poisoning
These details have moved to
RESEARCH on NAPHTHALENE HEALTH HAZARDS - separate article
Test for Naphthalene, Moth Flakes, Mothball Offgassing Level
This topic is now found
at NAPHTHALENE / PARABENTIOL OFF-GAS TESTING
Termites & Other Natural Sources of Naphthalene: Possible Hazards to Humans
This topic has moved to its own page at
TERMITES & NAPHTHALENE HAZARDS - separate article
...
Reader Comments, Questions & Answers About The Article Above
Below you will find questions and answers previously posted on this page at its page bottom reader comment box.
Reader Q&A - also see RECOMMENDED ARTICLES & FAQs
On 2020-07-03 - by (mod) - Is sunlight enough to neutralize naphthalene?
Carla
"Neutralize" suggests a chemical reaction of which I cannot offer an opinion in this case. You can read some details in the article above - note the use of the term sublime or sublimation.
Any household cleaner or upholstery cleaner will remove some but often not all of the mothball odor, as can professional cleaning.
I'm not sure I've convinced you to review the material in this article and its companions. My best suggestions are in the articles above. Please take a look and let me know what you think.
Often fresh air and sunlight will speed off-gassing of mothball odors.
On 2020-07-03 by Carla De Vries - neutralize naphthalene?
Is sunlight enough to neutralize naphthalene?
my daughter is concerned with toxicity due to dermal transmission (her children touching the upholstery and getting pesticide poisoning), not odor.
What product, if any, cleans away naphthalene?
On 2020-06-29 - by (mod) - ok to store oriental rugs rolled up with mothballs ?
 Scott
Scott
An "overdose" of mothball odors can be very difficult to remove from porous materials including oriental rugs stored in the intense conditions you are considering.
I'd defer to advice from a rug expert, such as those at George Washington University GWU - where experts warn:
Most moth and carpet beetle infestations appear in stored rugs. The source of the infestation may have occurred earlier when the piece was being used, but in the quiet, dark surroundings of storage the infestation may quickly grow. - retrieved 2020/06/29 original source: George Washington University Museum, museum.gwu.edu/storing-oriental-carpets
Left alone I'd probably have the rugs professionally cleaned now, then when absolutely dry, rolled in clear polyethylene or polypropylene and stored in a cool, dry place; the wrapping can be mothproof without dosing the rugs themselves. If there were space you might store rugs flat, separated by acid-free paper.
Illustrated here: U-Haul (and others) sell plastic bags intended for rug storage.
Watch out: don't store oriental rugs for a long time folded - the creases can damage the fibres and can also be hard to remove later.
Watch out: while experts agree on plastic as a storage wrapper - it has the advantage of being moth resistant - take care that your particular roll of polyethylene does not have its own horrible smell. Varying significantly by source as well as, I opine, production run, some poly off-gasses a terrible and hard-to-remove smell that can be sickening. Sniff first, use second.
On 2020-06-29 by Scott
Thanks for your wonderfully-informative site. I just discovered a moth infestation in a back room with about 10 oriental rugs rolled-up for storage [lots of wool and silk threads]. The rugs were valuable at one time, but that market shrank about 15 years ago -- There are probably 10x as many sellers as buyers ...
Nobody wants to even look at them with moth infestation, so I'm tempted to load them in heavy plastic bags with mothballs for a few months. The question then becomes: is it realistic to hope that the rugs can eventually be cleaned in a way that eliminates the mothball odor ?
Thanks for any guidance you can offer on this !
On 2019-08-28 - by (mod) - is mothball odor harmful?
Bonnie
The possible health effects of exposure to mothballs or mothball odor (naphthalene) are discussed on this page - a good place to look is at
- HEALTH EFFECTS of EXPOSURE to MOTHBALLS
- NAPHTHALENE POISONING SYMPTOMS
Please check that out and don't hesitate to ask any follow-up questions needed.
On 2019-08-28 by Bonnie Hughes
The smell of moth balls is exceptionally strong in the hallway of my apartment complex...is it harmful
On 2019-02-18 - by (mod) - minimum mothball odor exposure to cause health problems
Unfortunately there's not a simple right answer to it the way you phrase it.
That's because with any environmental hazard the time duration of exposure is only part of the equation when assessing the hazard level or risk. It's the concentration level that is critical as well. In the article above we state
the sublimation rate of mothballs depends on the relative naphthalene gas or para-dichlorobenzene vapor pressure difference between the thin layer of air at the surface of the mothball and the surrounding air.
And that's just one of several scientific details. Indoor air movement, temperature, and humidity level would also be factors. So no one based on an e-text can tell you how long a generic exposure to mothball odors would have to occur for anyone to suffer any ill effect.
Nevertheless in this is subjective opinion I can state that I certainly haven't seen research claiming that a brief exposure has produced lasting health effects.
I'll do some further research to see if we can put some additional numbers on this interesting question.
On 2019-02-18 by Sarah -
What is the minimum time of exposure to a very very strong smell of moth balls to potientially cause health problems to young children or an unborn baby. I know there is a certain type of anemia it can cause.
And again, smell was overwhelming, not sure how many moth balls or where in the house, but obviously a lot to cause such a strong odor through the whole house in such a strong form
...
Continue reading at TERMITES & NAPHTHALENE HAZARDS - mothball odors may not be from mothballs! - or select a topic from the closely-related articles below, or see the complete ARTICLE INDEX.
Or see MOTHBALL ODOR HAZARD FAQs questions & answers about mothballs posted originally at this article.
Or see these
Recommended Articles
- ENDOCRINE DISRUPTERS at BUILDINGS
- INSECT INFESTATION / DAMAGE - home
- MOTHS, MOTHBALL ODORS - home
- HEALTH EFFECTS of EXPOSURE to MOTHBALLS
- REDUCE INDOOR EXPOSURE to MOTHBALL CHEMICALS
- GET RID of MOTHBALL ODORS
- LIFE EXPECTANCY of MOTHBALLS
- MOTHBALL ODOR IMPACT on REAL ESTATE SALES
- TERMITES & NAPHTHALENE HAZARDS
- NAPHTHALENE POISONING SYMPTOMS
- NAPHTHALENE HEALTH HAZARD RESEARCH
- NAPHTHALENE / PARABENTIOL OFF-GAS TESTING
- ODORS GASES SMELLS, DIAGNOSIS & CURE - home
- OZONE AIR PURIFIER WARNINGS
- PESTICIDE EXPOSURE HAZARDS
- POWDER POST BEETLES & CARPET BEETLES - control, fumigation
- REPELLENTS for ANIMAL PESTS
- SMELL PATCH TEST to FIND ODOR SOURCE
- TERMITES & NAPHTHALENE HAZARDS
Suggested citation for this web page
MOTHS, MOTHBALL ODORS at InspectApedia.com - online encyclopedia of building & environmental inspection, testing, diagnosis, repair, & problem prevention advice.
Or see this
INDEX to RELATED ARTICLES: ARTICLE INDEX to BUILDING ODOR DIAGNOSIS & CURE
Or use the SEARCH BOX found below to Ask a Question or Search InspectApedia
Or see
INDEX to RELATED ARTICLES: ARTICLE INDEX to INSECT DAMAGE
Or use the SEARCH BOX found below to Ask a Question or Search InspectApedia
Ask a Question or Search InspectApedia
Try the search box just below, or if you prefer, post a question or comment in the Comments box below and we will respond promptly.
Search the InspectApedia website
Note: appearance of your Comment below may be delayed: if your comment contains an image, photograph, web link, or text that looks to the software as if it might be a web link, your posting will appear after it has been approved by a moderator. Apologies for the delay.
Only one image can be added per comment but you can post as many comments, and therefore images, as you like.
You will not receive a notification when a response to your question has been posted.
Please bookmark this page to make it easy for you to check back for our response.
IF above you see "Comment Form is loading comments..." then COMMENT BOX - countable.ca / bawkbox.com IS NOT WORKING.
In any case you are welcome to send an email directly to us at InspectApedia.com at editor@inspectApedia.com
We'll reply to you directly. Please help us help you by noting, in your email, the URL of the InspectApedia page where you wanted to comment.
Citations & References
In addition to any citations in the article above, a full list is available on request.
- World Health Organization, The WHO RECOMMENDED CLASSIFICATION of PESTICIDES by Hazard and Guidelines to Classification 2009 [PDF], retrieved 2017/07/.27, original source: http://www.who.int/ipcs/publications/pesticides_hazard_2009.pdf?ua=1
- [2] Thomas M. Riddick, "Controlling Taste, Odor and Color With Free Residual Chlorination", Journal (American Water Works Association) Vol. 43, No. 7 (JULY 1951), pp. 545-552, American Water Works Association, Article Stable URL: http://www.jstor.org/stable/41236445
- [8] Black, Judy. Fabric and Museum Pests. In Mallis Handbook of Pest Control, 9th edition, S.A. Hedges and D. Moreland, eds. GIE Media, Cleveland, OH, 2004, pp. 581 –623.
- In addition to citations & references found in this article, see the research citations given at the end of the related articles found at our suggested
CONTINUE READING or RECOMMENDED ARTICLES.
- Carson, Dunlop & Associates Ltd., 120 Carlton Street Suite 407, Toronto ON M5A 4K2. Tel: (416) 964-9415 1-800-268-7070 Email: info@carsondunlop.com. Alan Carson is a past president of ASHI, the American Society of Home Inspectors.
Thanks to Alan Carson and Bob Dunlop, for permission for InspectAPedia to use text excerpts from The HOME REFERENCE BOOK - the Encyclopedia of Homes and to use illustrations from The ILLUSTRATED HOME .
Carson Dunlop Associates provides extensive home inspection education and report writing material. In gratitude we provide links to tsome Carson Dunlop Associates products and services.











